Drug Discovery Services
Vivonics is a contract research organisation established in 2015. We specialise in assessing the effects of potential new medicines on the function of vital organs.
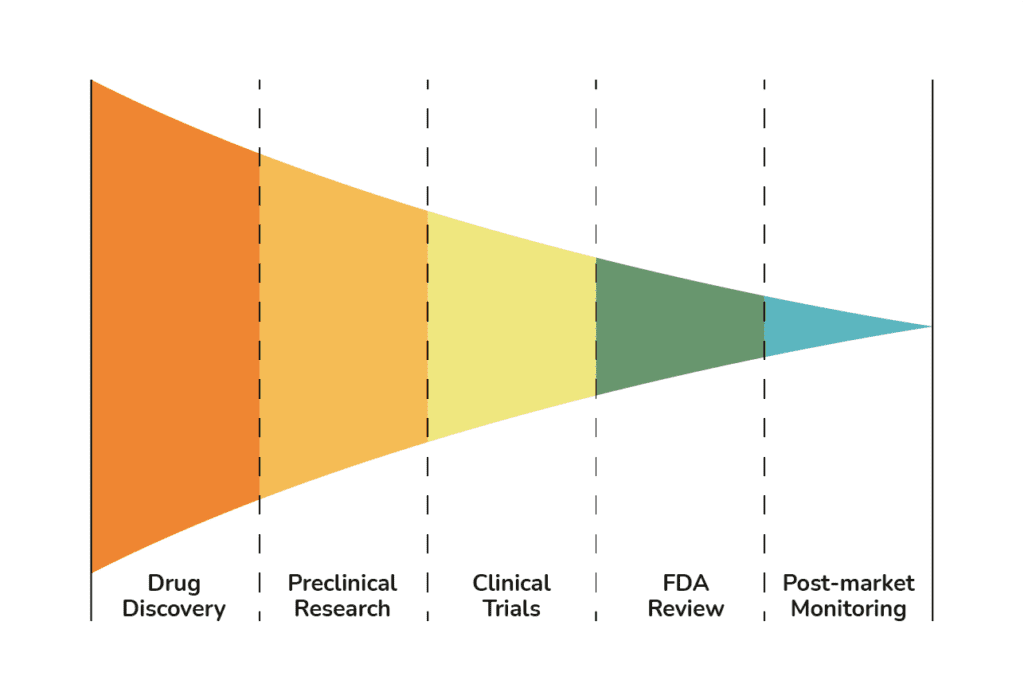
Our Services
Using our validated in vivo assays, we investigate the potential safety liabilities of your lead molecules and deliver high-quality data and interpretation back to your project team to help inform future decisions and strategies.

latest News

How is animal research regulated in the UK?
Welfare is the at forefront of studies conducted here at Vivonics but how is animal research regulated in the UK?

Animal Welfare – Vivonics Replacement, Reduction, Refinement Policy
Vivonics replacement, reduction and refinement policy is aimed at keeping animal welfare a top priority & improving our procedures.

Waveform analysis of blood pressure telemetry data
This method utilises all the numerical data available, replotting and revisualizing them in a manner that allows unique quantification of changes in waveform morphology and variability
Vital organ function, measured, analysed and interpreted

